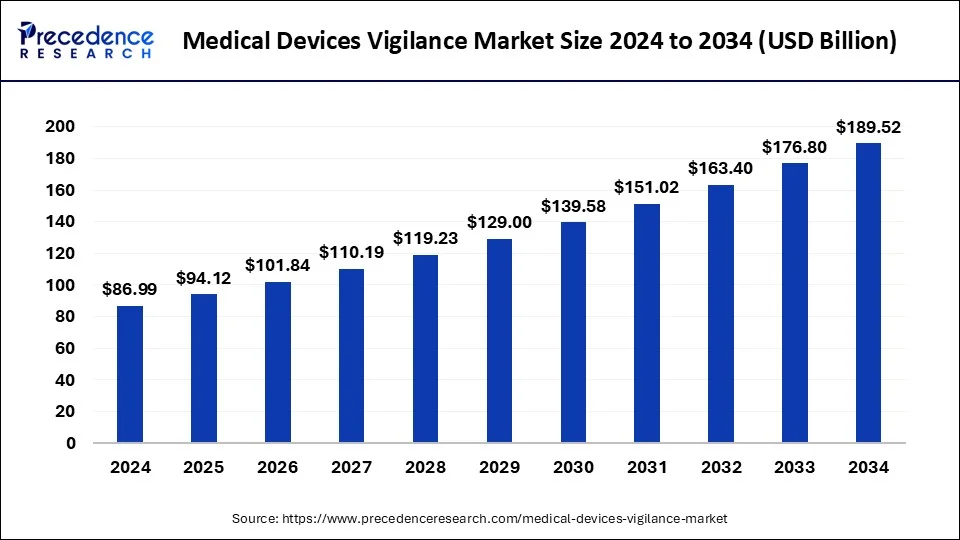
The global medical devices vigilance market size accounted for USD 80.40 billion in 2023 and is anticipated to grow around USD 176.80 billion by 2033, growing at a CAGR of 8.19% from 2024 to 2033.
Key Points
- The North America medical devices vigilance market size reached USD 24.12 billion in 2023 and is expected to attain around USD 53.04 billion by 2033.
- North America was estimated to hold a substantial market share of 34% in 2023.
- Asia Pacific is projected to witness rapid growth in the global market.
- By delivery mode, the on-demand segment accounted for the largest share of 81% in 2023.
- By delivery mode, the on-premises segment is expected to have steady growth over the forecast period.
- By application, the diagnostics segment held a substantial market share of 36% in 2023.
- By application, the research segment is expected to show lucrative growth over the forecast period.
- By end use, the clinical research organization segment held the highest market share of 42% in 2023.
- By end use, the business process outsourcing firms segment is expected to grow rapidly in the foreseeable period.
The medical devices vigilance market encompasses the monitoring and reporting of adverse events and safety concerns related to medical devices. This market plays a critical role in ensuring the safety and efficacy of medical devices, facilitating prompt actions and regulatory compliance. Medical devices vigilance systems include reporting systems, data analysis tools, and communication channels for healthcare providers, manufacturers, and regulatory authorities.
Get a Sample: https://www.precedenceresearch.com/sample/4120
Growth Factors
The growth of the medical devices vigilance market is driven by an increasing focus on patient safety and regulatory compliance. There is a rising awareness of the importance of monitoring medical device performance to detect potential risks and adverse events. Additionally, technological advancements such as data analytics and AI-powered tools are enabling more efficient and proactive monitoring of medical devices, contributing to the market’s growth.
Region Insights
Regionally, North America is expected to dominate the medical devices vigilance market due to its strong regulatory framework and established healthcare infrastructure. Europe follows closely, benefiting from stringent safety regulations and active monitoring systems. The Asia-Pacific region is also expected to experience significant growth due to increasing investment in healthcare infrastructure and the rising adoption of advanced medical devices.
Medical Devices Vigilance Market Scope
| Report Coverage | Details |
| Growth Rate from 2024 to 2033 | CAGR of 8.19% |
| Global Market Size in 2023 | USD 80.40 Billion |
| Global Market Size in 2024 | USD 86.99 Billion |
| Global Market Size by 2033 | USD 176.80 Billion |
| Largest Market | North America |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Segments Covered | By Delivery Mode, By Application, and By End-user |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Medical Devices Vigilance Market Dynamics
Drivers
Key drivers of the medical devices vigilance market include regulatory mandates that require manufacturers to establish vigilance systems and report adverse events. Additionally, the growing complexity of medical devices and their widespread use in various healthcare settings necessitate robust monitoring systems. Increasing patient awareness and advocacy for safety also contribute to market growth.
Opportunities
Opportunities in the medical devices vigilance market include the development of advanced data analytics and AI-driven tools to enhance monitoring capabilities. Collaboration between manufacturers, healthcare providers, and regulatory authorities can lead to improved vigilance practices and shared knowledge. Expansion into emerging markets with growing healthcare needs presents another opportunity for market growth.
Challenges
Challenges in the medical devices vigilance market include the complexity of data collection and analysis from various sources and devices. Ensuring compliance with diverse international regulations can be challenging for manufacturers operating in multiple regions. Additionally, balancing the need for vigilance with the risk of over-reporting or misinterpretation of data can pose challenges in maintaining effective systems.
Read Also: Tire Pyrolysis Oil Market Size to Grow USD 567.90 Mn by 2033
Medical Devices Vigilance Market Recent Developments
- In June 2022, Italy instituted substantial changes in national regulations on medical device vigilance in accordance with the procedures by European Regulations. 2017/475 for medical devices and 2017/476 for in vitro diagnostics.
Medical Devices Vigilance Market Companies
- ZEINCRO
- AssurX Inc.
- Sparta System
- Oracle Corporation
- Xybion Corporation
- Sarjen Systems Pvt. Ltd.
- MDI Consultants, Inc.
- AB-Cube
- Laerdal Medical.
- Omnify Software, Inc.
Segments Covered in the Report
By Delivery Mode
- On-demand
- On-premise
By Application
- Diagnostics
- Therapeutics
- Surgical
- Research
By End-user
- Clinical Research Organizations (CROs)
- Business Process Outsourcing (BPO)
- Original Equipment Manufacturers (OEM)
- Other End-users
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
Blog: https://www.dailytechbulletin.com/
Blog: https://www.autoindustrybulletin.com/
