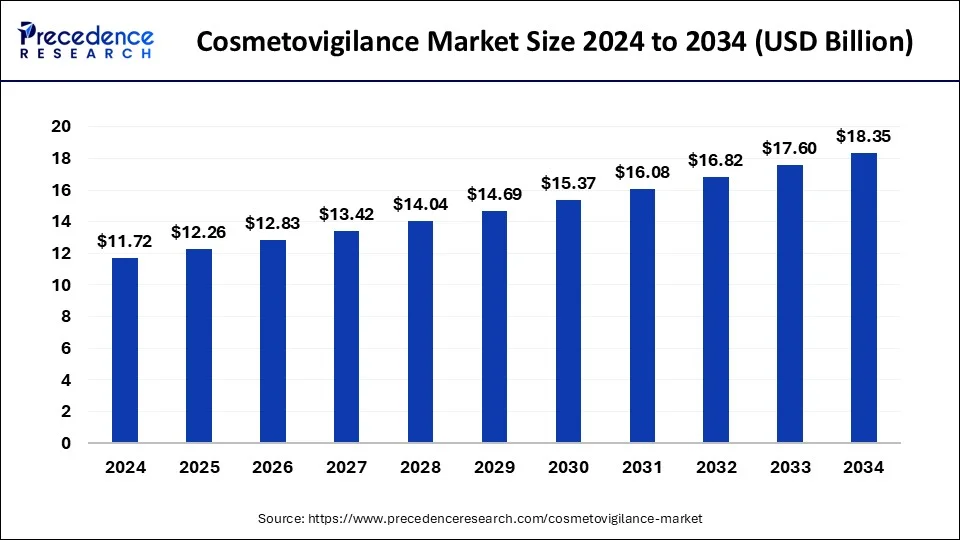
The global cosmetovigilance market size accounted for USD 11.20 billion in 2023 and is projected to rake around USD 17.60 billion by 2033, growing at a CAGR of 4.62% from 2024 to 2033.
Key Points
- Europe has captured highest revenue share in cosmetovigilance market in 2023 with 37.8%.
- By service type, the post-marketing services market segment accounted for the dominant share of the market in 2023 with 64%.
- By service type, the pre-marketing services segment is expected to witness considerable growth in the global market over the forecast period.
- By categories, the skincare segment held the largest share of around 33% in 2023.
- By service provider, the contract outsourcing segment has accounted largest revenue share of 60% in 2023.
- By phase type, the Phase IV segment has held revenue share of 76% in 2023.
The cosmetovigilance market encompasses the monitoring, assessment, and management of adverse events and risks associated with cosmetic products. Similar to pharmacovigilance in the pharmaceutical industry, cosmetovigilance focuses on ensuring the safety and efficacy of cosmetics, skincare, and personal care products. With the growing demand for cosmetics worldwide, fueled by changing consumer preferences, increased awareness of beauty and wellness, and evolving regulatory standards, cosmetovigilance plays a crucial role in safeguarding public health and consumer confidence.
Get a Sample: https://www.precedenceresearch.com/sample/4062
Growth Factors:
Several factors drive the growth of the cosmetovigilance market. Firstly, the expanding global cosmetics industry, driven by rising disposable incomes, urbanization, and social media influence, has led to a proliferation of cosmetic products in the market. As consumers demand more diverse and innovative beauty solutions, the need for effective cosmetovigilance measures to monitor product safety becomes paramount.
Moreover, stringent regulatory requirements and increasing scrutiny from regulatory authorities compel cosmetics manufacturers to invest in cosmetovigilance systems and processes to comply with safety standards and regulations. Regulatory bodies such as the FDA in the United States, the European Commission in Europe, and the CFDA in China impose strict guidelines for the safety assessment and post-market surveillance of cosmetic products, driving demand for cosmetovigilance services.
Additionally, growing consumer awareness and concern regarding product safety, ingredient transparency, and adverse reactions contribute to the demand for cosmetovigilance. High-profile incidents involving product recalls, adverse reactions, and controversies over harmful ingredients have underscored the importance of cosmetovigilance in ensuring consumer trust and brand reputation.
Region Insights:
The cosmetovigilance market exhibits regional variations influenced by factors such as regulatory frameworks, consumer preferences, and market maturity.
In North America, stringent regulatory oversight by agencies such as the FDA and Health Canada drives the adoption of comprehensive cosmetovigilance practices among cosmetics manufacturers. The region’s sophisticated healthcare infrastructure and consumer awareness further contribute to the demand for cosmetovigilance services.
In Europe, the European Union’s robust regulatory framework, including the Cosmetic Regulation (EC) No 1223/2009, mandates stringent safety assessment and post-market surveillance of cosmetic products. As a result, cosmetovigilance activities are well-established, with companies investing in pharmacovigilance systems and processes to comply with regulatory requirements.
In Asia-Pacific, rapid urbanization, increasing disposable incomes, and changing beauty trends fuel the growth of the cosmetics market. Countries such as China, Japan, and South Korea are key markets for cosmetics, driving demand for cosmetovigilance services to ensure product safety and regulatory compliance.
Cosmetovigilance Market Scope
| Report Coverage | Details |
| Global Market Size in 2023 | USD 11.20 Billion |
| Global Market Size in 2024 | USD 11.72 Billion |
| Global Market Size by 2033 | USD 17.60 Billion |
| Growth Rate from 2024 to 2033 | CAGR of 4.62% |
| Largest Market | Europe |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Segments Covered | By Service Type, By Categories, By Phase Type, and By Service Provider |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Cosmetovigilance Market Dynamics
Drivers:
Several drivers propel the cosmetovigilance market forward. Firstly, the increasing globalization of the cosmetics industry, coupled with cross-border trade and e-commerce, necessitates harmonized regulatory standards and cosmetovigilance practices to ensure consistent product safety and quality worldwide.
Furthermore, advancements in technology, such as artificial intelligence, data analytics, and digital platforms, enable more efficient and proactive cosmetovigilance monitoring and risk assessment. Automated adverse event reporting systems, real-time data analysis, and predictive modeling enhance the detection and management of safety signals, facilitating timely interventions and risk mitigation measures.
Moreover, collaborations between regulatory authorities, industry stakeholders, and healthcare professionals play a vital role in strengthening cosmetovigilance efforts. Information sharing, training programs, and collaborative research initiatives enhance awareness, competence, and capacity-building in cosmetovigilance across the value chain.
Opportunities:
The cosmetovigilance market presents various opportunities for stakeholders to capitalize on emerging trends and address evolving challenges. Firstly, the integration of digital technologies, such as mobile apps, wearable devices, and online forums, empowers consumers to report adverse events and access product information in real-time, fostering transparency and accountability in the cosmetics industry.
Moreover, the growing demand for natural, organic, and sustainable cosmetics presents an opportunity for cosmetovigilance to address safety concerns related to natural ingredients, allergens, and environmental impact. Robust safety assessment protocols, ingredient testing, and risk communication strategies can enhance consumer confidence in green beauty products.
Furthermore, the convergence of cosmetovigilance with other disciplines such as medical aesthetics, nutricosmetics, and personalized skincare opens new avenues for research, innovation, and collaboration. By leveraging synergies between cosmetovigilance and adjacent fields, stakeholders can enhance product safety, efficacy, and consumer satisfaction.
Challenges:
Despite the opportunities, the cosmetovigilance market faces several challenges that need to be addressed to ensure its effectiveness and sustainability. Firstly, the lack of standardized reporting systems, terminology, and data interoperability hampers the harmonization and exchange of cosmetovigilance information across regions and stakeholders.
Moreover, the dynamic nature of the cosmetics industry, characterized by rapid product innovation, ingredient substitution, and supply chain complexities, poses challenges for cosmetovigilance surveillance and risk assessment. Timely identification and assessment of emerging safety concerns require agile and adaptive cosmetovigilance systems capable of keeping pace with industry developments.
Furthermore, resource constraints, including limited funding, expertise, and infrastructure, may hinder the implementation of robust cosmetovigilance programs, particularly in emerging markets and small-to-medium enterprises. Capacity-building initiatives, public-private partnerships, and regulatory support are essential to address these challenges and strengthen cosmetovigilance capabilities globally.
Read Also: IT Services Market Size to Rise USD 2.80 Trillion By 2033
Recent Developments
- In December 2023, Biofrontera AG, an international biopharmaceutical company, announced that its wholly-owned subsidiary Biofrontera Bioscience GmbH has received a notice of allowance for the application “Photodynamic therapy comprising two light exposures at different wavelengths by the US patent office USPTO. This patent protects several innovations relating to a new illumination method to treat dermatological skin diseases with Photodynamic Therapy (PDT).
- In June 2023, Anju Software and ClinChoice partnered to enhance eClinical solutions for clinical research. ClinChoice’s acquisition of Cromsource led to the collaboration, enabling the delivery of adaptable and cost-efficient eClinical solutions, including TrialMaster, for streamlined clinical trial data management and regulatory compliance.
Cosmetovigilance Market Companies
- Poseidon CRO
- AxeRegel
- PharSafer
- AB Cube
- Aixial Group
- Di Renzo
- Pharmathen
- Skill Pharma
- OC Vigilance
- Preclinical
- safety
- ZEINCRO Group
- FMD K&L
- POSEIDON CRO
- MSL Solution Providers
- Cliantha
- Pure Drug Safety
- KMJ pharma sp. z o.o.
- TheraSkin
- CORONIS Research SA
- PHARMALEX GMBH
- Bioclinica
- Tecnimede Group
- Sciformix
Segments Covered in the Report
By Service Type
- Pre-marketing Services
- Clinical Safety Testing
- Document Writing
- Risk Management
- Post-marketing Services
- Case Intake
- Case Triage
- Data Entry & Acquisition
- Tracking & Reporting
By Categories
- Skincare
- Makeup
- Haircare
- Perfumes and deodorants
- Hair colorants
- Others
By Phase Type
- Pre-clinical
- Phase I
- Phase II
- Phase III
- Phase IV
By Service Provider
- In-house
- Contract Outsourcing
- Contract Research Organizations (CROs)
- Business Process Outsourcing Organizations (BPOs)
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
Blog: https://www.dailytechbulletin.com/
Blog: https://www.autoindustrybulletin.com/
